Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)
GENERIC NAME(S): Upadacitinib
OTHER NAME(S): Rinvoq Tablet, Extended Release 24 Hr
Upadacitinib may lower your ability to fight infections. This may make you more likely to get a serious (rarely fatal) infection or make any infection you have worse. The most common serious infections include pneumonia and skin infections. The risk for infections may be higher if you also take other drugs that suppress the immune system (such as methotrexate, corticosteroids). Tell your doctor right away if you have any signs of infection (such as a sore throat that doesn't go away, fever, chills, cough, non-healing skin sores, painful/frequent urination, white patches in your mouth or on your tongue).
You should have a tuberculosis (TB) skin test before and during treatment with this medication. Tell your doctor if you have been near someone with tuberculosis or have lived or traveled in areas where certain fungal infections (such as coccidioidomycosis, histoplasmosis) are common. These areas include the Ohio and Mississippi River valleys and the southwestern United States.
Though it is very unlikely to occur, there may be a risk of developing cancer (such as lymphoma, skin cancer, lung cancer) with this medication. Your risk may be higher if you are a current or past smoker. Protect your skin from the sun. Avoid prolonged sun exposure, tanning booths, and sunlamps. Use a sunscreen and wear protective clothing when outdoors. Tell your doctor right away if you develop symptoms such as fever or cough that doesn't go away, wheezing, unusual lumps/growths, unexplained weight loss, night sweats, change in appearance or size of moles, or unusual skin changes.
Upadacitinib may rarely cause serious (possibly fatal) blood clots in the lungs or legs, or clots that cause a stroke or heart attack. You may be at an increased risk for blood clots if you are a current or past smoker, or are 50 years of age or older and have at least one risk factor for heart disease. Discuss the risks and benefits of treatment with your doctor. Get medical help right away if you develop symptoms of blood clots, such as shortness of breath/rapid breathing, chest/jaw/left arm pain, unusual sweating, confusion, sudden dizziness/fainting, pain/swelling/warmth in the groin/calf, sudden/severe headaches, trouble speaking, weakness on one side of the body, or sudden vision changes.
Who should not take Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)?
Show MoreUses
Upadacitinib is used to treat certain types of arthritis (such as psoriatic arthritis, rheumatoid arthritis, axial spondyloarthritis). It helps decrease pain, tenderness, and swelling in the joints. Upadacitinib is also used to treat a skin condition called atopic dermatitis. It reduces swelling, itching, and redness in the skin. This medication may also be used to treat certain bowel diseases (Crohn's disease, ulcerative colitis). It helps to reduce symptoms such as diarrhea and stomach pain.
How to use Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)
Read the Medication Guide and, if available, the Patient Information Leaflet provided by your pharmacist before you start taking upadacitinib and each time you get a refill. If you have any questions, ask your doctor or pharmacist.
Take this medication by mouth with or without food as directed by your doctor, usually once daily. Do not crush, chew, or split the tablets. Doing so can release all of the drug at once, increasing the risk of side effects.
Use this medication regularly to get the most benefit from it. To help you remember, take it at the same time each day.
The dosage is based on your medical condition, response to treatment, age, and other medications you may be taking. Be sure to tell your doctor and pharmacist about all the products you use (including prescription drugs, nonprescription drugs, and herbal products).
Do not increase your dose or use this drug more often or for longer than prescribed. Your condition will not improve any faster, and your risk of side effects will increase.
Avoid eating grapefruit or drinking grapefruit juice while using this medication unless your doctor or pharmacist says you may do so safely. Grapefruit can increase the chance of side effects with this medicine. Ask your doctor or pharmacist for more details.
Tell your doctor if your condition does not get better or if it gets worse.
Side Effects
See also Warning section.
Nausea or headache may occur. If either of these effects lasts or gets worse, tell your doctor or pharmacist promptly.
You may rarely see a tablet or partial tablets in your stool. This may occur if you have certain intestinal disorders (such as ileostomy, colostomy). Tell your doctor right away if you see tablets in your stool.
People using this medication may have serious side effects. However, you have been prescribed this drug because your doctor has judged that the benefit to you is greater than the risk of side effects. Careful monitoring by your doctor may decrease your risk.
Tell your doctor right away if you have any serious side effects, including: signs of anemia (such as unusual tiredness, pale skin, fast heartbeat), signs of liver disease (such as nausea/vomiting that doesn't stop, loss of appetite, stomach/abdominal pain, yellowing eyes/skin, dark urine), signs of a serious tear in the stomach or intestinal wall (such as severe stomach/abdominal pain that doesn't go away, fever, severe dizziness, fainting, severe nausea/vomiting).
Get medical help right away if you have any very serious side effects, including: sudden vision changes (such as blurred vision, seeing flashes of light).
A very serious allergic reaction to this drug is rare. However, get medical help right away if you notice any symptoms of a serious allergic reaction, including: rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist.
In the US -
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
In Canada - Call your doctor for medical advice about side effects. You may report side effects to Health Canada at 1-866-234-2345.
Precautions
See also Warning section.
Before taking upadacitinib, tell your doctor or pharmacist if you are allergic to it; or if you have any other allergies. This product may contain inactive ingredients, which can cause allergic reactions or other problems. Talk to your pharmacist for more details.
Before using this medication, tell your doctor or pharmacist your medical history, especially of: blood clots (such as in the lungs, legs), blood/bone marrow problems (such as anemia), cancer, heart problems (such as previous heart attack), liver disease (such as hepatitis B or C), past/recent/current infections (such as tuberculosis, herpes zoster, HIV infection), current or past smoking, stomach/intestinal problems (such as ulcers, diverticulitis), stroke, kidney disease.
Upadacitinib can make you more likely to get infections or may make current infections worse. Stay away from anyone who has an infection that may easily spread (such as chickenpox, COVID-19, measles, flu). Talk to your doctor if you have been exposed to an infection or for more details.
Tell your health care professional that you are using upadacitinib before having any immunizations/vaccinations. Avoid contact with people who have recently received live vaccines (such as flu vaccine inhaled through the nose).
Before having surgery, tell your doctor or dentist about all the products you use (including prescription drugs, nonprescription drugs, and herbal products).
Older adults may be at greater risk for side effects while using this drug.
Tell your doctor if you are pregnant or plan to become pregnant. You should not become pregnant while using upadacitinib. Upadacitinib may harm an unborn baby. Your doctor should order a pregnancy test before you start this medication. Ask about reliable forms of birth control while using this medication and for 4 weeks after the last dose. If you become pregnant, talk to your doctor right away about the risks and benefits of this medication.
It is unknown if this medication passes into breast milk. Because of the possible risk to the infant, breastfeeding is not recommended while using this medication and for 6 days after the last dose. Consult your doctor before breastfeeding.
Interactions
See also How to Use section.
Drug interactions may change how your medications work or increase your risk for serious side effects. This document does not contain all possible drug interactions. Keep a list of all the products you use (including prescription/nonprescription drugs and herbal products) and share it with your doctor and pharmacist. Do not start, stop, or change the dosage of any medicines without your doctor's approval.
Other medications can affect the removal of upadacitinib from your body, which may affect how upadacitinib works. Examples include rifampin, phenytoin, among others.
Overdose
If someone has overdosed and has serious symptoms such as passing out or trouble breathing, call 911. Otherwise, call a poison control center right away. US residents can call their local poison control center at 1-800-222-1222. Canada residents can call a provincial poison control center.
Notes
Do not share this medication with others.
Lab and/or medical tests (such as liver function, complete blood count, cholesterol levels, skin exams) should be done while you are taking this medication. Keep all medical and lab appointments.
Missed Dose
If you miss a dose, take it as soon as you remember. If it is near the time of the next dose, skip the missed dose. Take your next dose at the regular time. Do not double the dose to catch up.
Storage
Store in original bottle in the refrigerator or at room temperature away from light and moisture. Do not store in the bathroom. Keep all medications away from children and pets.
Do not flush medications down the toilet or pour them into a drain unless instructed to do so. Properly discard this product when it is expired or no longer needed. Consult your pharmacist or local waste disposal company.Information last revised November 2023. Copyright(c) 2024 First Databank, Inc.
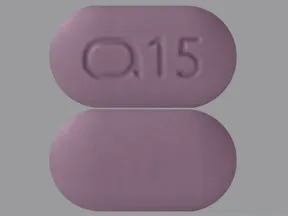


- color
- purple
- shape
- oblong
- imprint
- logo and 15

- color
- purple
- shape
- oblong
- imprint
- logo and 15
Drug Survey
Are you taking Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)?
Are you considering switching to Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)?
How satisfied are you with the results?
Are you planning to see a doctor about switching your medication?
How long have you been taking Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)?
Are you planning to see a doctor about switching your medication?
Thanks for taking our survey!
Recommended For You
Find a doctor near youMore Resources for Rinvoq ER 15 Mg Tablet,Extended Release Antirheumatic Agents (Biologic DMARDS)
You Might Also Like
Find More Drugs Used to Treat these Conditions
- ulcerative colitis, an inflammatory condition of the intestines
- Crohn's disease
- rheumatoid arthritis
- psoriasis associated with arthritis
- a type of allergy that causes red and itchy skin called atopic dermatitis
- ankylosing spondylitis, a rheumatic disease causing pain and stiffness in backbone
- non-radiographic axial spondyloarthritis
